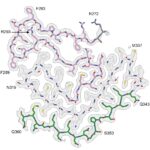
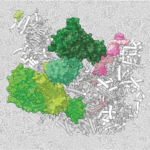
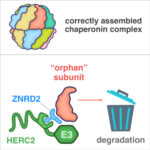
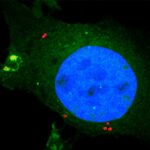
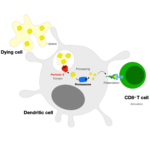
Sean Munro’s group in the LMB’s Cell Biology Division have investigated the potential biological significance of conserved genes of unknown function by developing an Unknome database to systematically analyse their identification and characterisation.