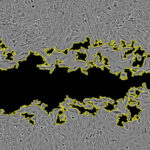
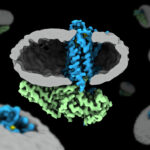
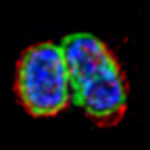
Multiple ribosomes speed along an mRNA to translate the genetic code into proteins. Szymon Juszkiewicz and Manu Hegde now find that when ribosomes collide, cells stop new ribosomes from starting translation and recruit a factor that clears the collision.