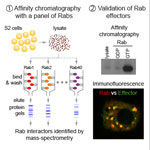
A research team from the LMB’s Cell Biology division, working with colleagues from the Structural Studies division, has revealed how cells are able to find and tag for degradation the partially synthesised proteins generated when ribosomes occasionally stall.
Cells make more than a hundred thousand new proteins every minute. Once in a while, one of the ribosomes making these proteins stalls, leaving an unfinished protein fragment.