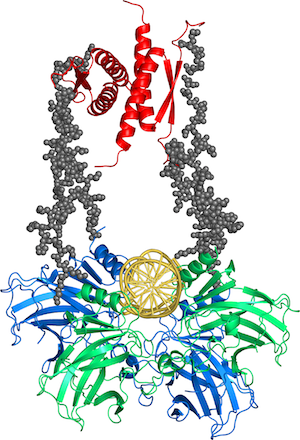
Cancer is a disease of mutation, and the most commonly mutated protein by far is the tumour suppressor p53. This protein, sometimes called the "Guardian of the Genome", does not only lead the major defence of the cell against cancer but also has a wide range of roles in the cell cycle, from fertility to senescence. It has a very complicated structure, comprised of two folded and three intrinsically disordered domains in each of its monomers that associate to form dimers and tetramers. It is a major hub protein and interacts with a host of other proteins involved in the cell cycle. We discovered that about 30% of oncogenic mutants of p53 are just temperatures sensitive and can, in theory, be rescued by small molecules.
Structural Biology and Drug Discovery
| Curriculum
vitae |
| Centre for Protein Engineering (CPE) |
| Photos
of
Group Members and CPE from 1985-2010 |
Alan Fersht
MRC Laboratory of Molecular Biology
Francis Crick Avenue
Cambridge CB2 0QH
Email alan@mrc-lmb.cam.ac.uk