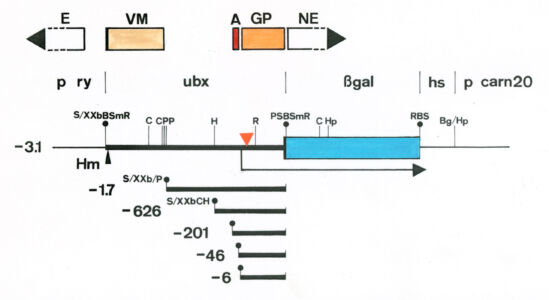
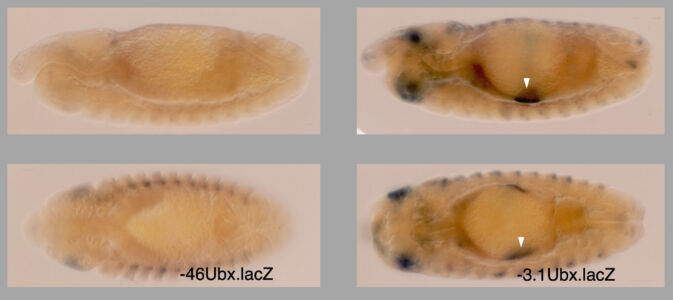
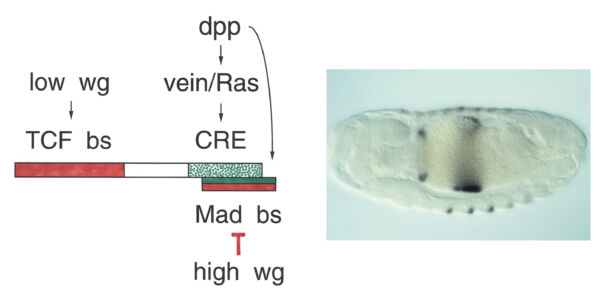
Having defined the cis-regulatory elements within HOX enhancers that respond to extracellular signals, we had the tools to identify the cis-acting factors that bound to these elements to respond to incoming signals. For Dpp, these factors were known to be the sequence-specific DNA-binding SMAD proteins, which turned out to bind to the Dpp-response element within the Ultrabithorax midgut enhancer. CREB and Fos also bind to this enhancer to respond to Vein signalling, but the DNA-binding factor responding to Wingless was unknown.
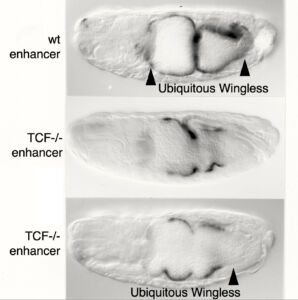
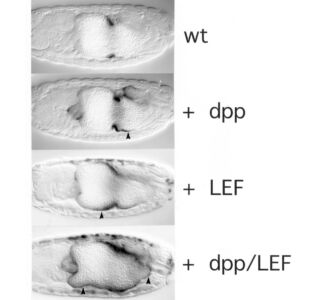
We surmised that this factor would bind directly to Armadillo (Drosophila beta-catenin), known to be the most downstream effector of the Wingless signalling cascade. We therefore used a yeast two-hybrid screen with activated Armadillo as bait to identify this transcription factor, but found a relative of the APC tumour suppressor instead (see below). However, we noticed that the Wnt-responsive elements in the Ultrabithoraxand labial enhancers contained multiple matches to the consensus binding site of TCF/LEF factors. These factors were discovered in mammalian T cells as sequence-specific DNA-binding proteins acting through an enhancer upstream of the T cell receptor-alpha gene. Unusually, these factors cannot activate transcription on their own but depend on cooperation with other transcription factors bound to the same enhancer, including CREB. We thus discovered that Drosophila TCF synergises with SMAD and CREB at the Ultrabithorax midgut enhancer to drive Ultrabithorax expression in the visceral mesoderm. The same three factors also co-operate with Dfos at the labial midgut enhancer to drive Labial expression in the midgut epithelium. Our work corroborated the principle of the context-dependence of TCF/LEF factors discovered by Waterman & Jones in 2001 through their studies of human LEF1.