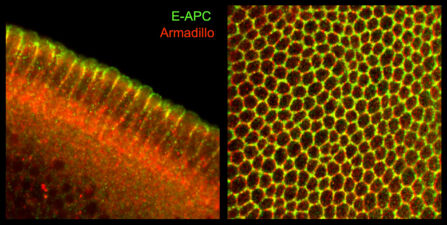
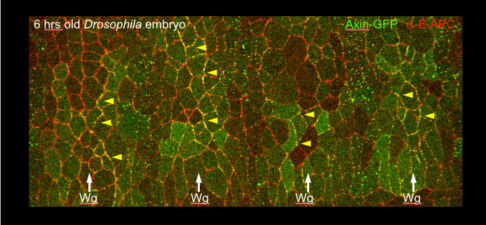
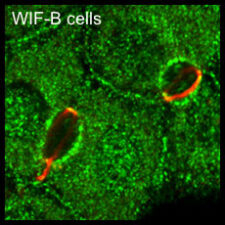
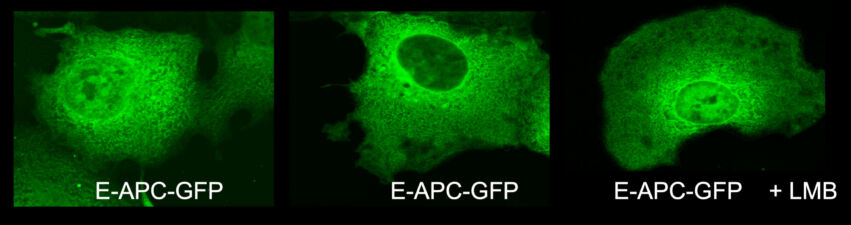
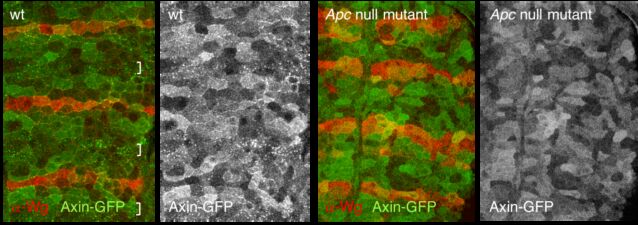
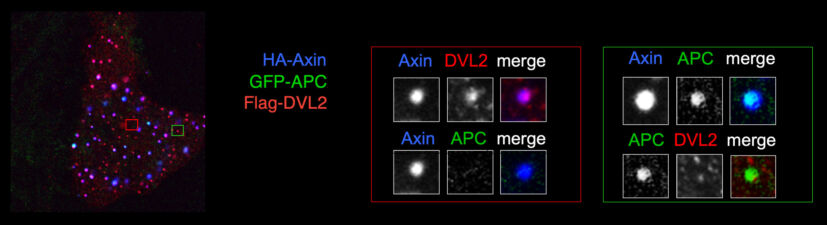
Our yeast two-hybrid screen based on activated Armadillo led us to discover a second Drosophila APC paralogue as an Armadillo-binding protein, which we called E-APC owing to its expressed in epithelia and association with E-cadherin and Armadillo at adherens junctions. E-APC (aka dAPC2) functions as an essential negative regulator of Armadillo, redundantly with its paralogue dAPC1 as shown by the Peifer group. Our evidence from flies and human epithelial cell models indicated that E-APC and its human counterpart, the APC tumour suppressor, promote shuttling of Armadillo/beta-catenin along actin filaments between the nuclear signalling and the cell adhesion pool. An interesting corollary is that APC proteins, like their Armadillo/beta-catenin binding partners, have dual functions in Wnt signalling and cellular adhesion.
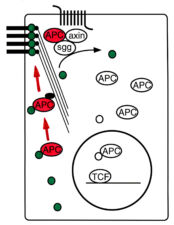
Support for this notion came from a point mutation (N175K) that we discovered in a genetic screen: N175 is a highly conserved glutamine in the Armadillo Repeat of E-APC predicted to contact its binding partners. In fly embryos, the N175K mutant E-APC protein is delocalised from adherens junctions and inactive in destabilising Armadillo. Its junctional association therefore seemed important for its function in the so-called beta-catenin destruction complex (or Axin degradasome, see next section) which earmarks beta-catenin/Armadillo for ubiquityation and proteasomal degradation in the absence of Wnt. Intriguingly, N175K also causes adhesion defects in multiple fly tissues, which indicated a function of E-APC in maintaining E-cadherin- and Armadillo-dependent cell adhesion.
In a yeast two-hybrid screen designed to identify human proteins binding to wild-type but not N175K APC, we identified a de-ubiquitylase (DUB) called Trabid. Trabid cleaves non-canonical ubiquitin chains, and APC is one of its substrates. Intriguingly, lack of Trabid function results in hyperactive APC and attenuated Wnt signalling in APC mutant cancer cells and reduces the intestinal tumour load of mice bearing Apc mutations. Loss of Trabid also causes defects of the peripheral actin cytoskeleton in human epithelial cells, though it remains to be seen whether this reflects a function of Trabid in promoting cell adhesion.