Acetylcholine receptor
The acetylcholine (ACh) receptor is a pentameric ion channel made from three similar and two identical polypeptide chains, arranged in a ring around a narrow membrane pore. ACh binds to this assembly at sites projecting far into the synaptic cleft, whereas the gate is located in the membrane pore more than 50Å away. How is the effect of ACh binding communicated almost instantly over such a large distance to open the gate and create an ion-conducting path?
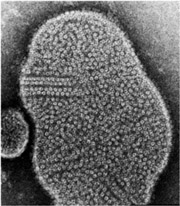
Postsynaptic membranes from the (muscle-derived) electric organ of the Torpedo ray provide an ideal source for structure-function studies probing this question. The isolated vesicles contain high concentrations of ACh receptors which, upon incubation in low salt solution, reorganise to form tubular crystals. Receptors in these tubes lie on a helical surface lattice, in the same packing arrangement and with similar curvature as is found at the synapse in situ.
Key publications:
Brisson, A. and Unwin, P.N.T. Tubular crystals of acetylcholine receptor. J. Cell Biol. 99, 1202-1211 (1984). (pdf)
Brisson, A. and Unwin, P.N.T. Quaternary structure of the acetylcholine receptor. Nature 315, 474-477 (1985). (pdf)
Unwin, N. The structure of ion channels in membranes of excitable cells. Neuron 3, 665-676 (1989). (pdf)