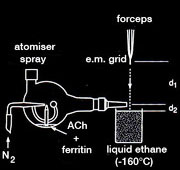
Spray-freeze-trapping
The plunge-freezing method can be combined with rapid mixing of spray droplets to capture transient conformations of proteins on the microscope grid. The two reactants (e.g. protein and ligand) are mixed briefly by coalesence of spray droplets containing one component with a thin, grid-supported aqueous film containing the other. Plunge-freezing then traps the transient state, which can be investigated later by cryo-microscopy.
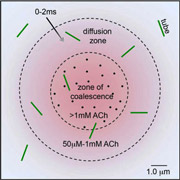
Spray-freeze-trapping was developed originally to obtain images of the acetylcholine receptor in the open-channel form. The ACh spray solution contained ferritin marker particles, and the activated ACh receptor tubes were identified by the presence of ferritin in the images. ACh-receptor reaction times were made <10ms to minimise ‘contamination’ by receptors converting to a desensitised form. In a later refinement of this technique, it was possible to identify tubes in the ‘diffusion zone’, located beyond the ferritin-delineated edges of the spreading droplets, thereby reducing reaction times to <2ms.
Key publications:
Berriman, J. and Unwin, N. Analysis of transient structures by cryo-microscpoy combined with rapid mixing of spray droplets. Ultramicroscopy 56, 241-252 (1994). (pdf)
Unwin, N. Acetylcholine receptor channel imaged in the open state. Nature 373, 37-43 (1995). (pdf)
Unwin, N. and Fujiyoshi, Y. Gating movement of acetylcholine receptor caught by plunge-freezing. J. Mol. Biol. 422, 617-634 (2012). (pdf)