 New research, from a team of scientists in the LMB’s Structural Studies Division and the Texas A&M Health Science Center, illustrates the molecular mechanism behind a fundamental cellular process.
New research, from a team of scientists in the LMB’s Structural Studies Division and the Texas A&M Health Science Center, illustrates the molecular mechanism behind a fundamental cellular process.
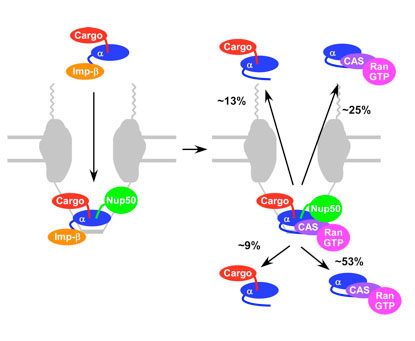
The research, published in PNAS, provides new insights into the way in which components of the nuclear protein transport machinery move through nuclear pores. Nuclear pores are large protein complexes that cross the nuclear envelope – the double membrane surrounding the eukaryotic cell nucleus. Nuclear pore complexes (NPCs) allow the transport of water-soluble molecules across the nuclear envelope. This transport includes RNA and ribosomes moving from nucleus to the cytoplasm and proteins, carbohydrates, signaling molecules and lipids moving into the nucleus.
Lead researcher Changxia Sun, from Siegfried Musser’s lab at Texas A&M, explains the team’s approach: “Nucleocytoplasmic transport requires the orchestrated assembly and disassembly of cargo-carrier complexes. By using single-molecule FRET (fluorescence energy transfer) and particle tracking we were able to visualise the choreography of assembly and disassembly at nuclear pores.”
LMB Group Leader Murray Stewart continues: “The research showed that although key interactions between components are reversible and multiple outcomes are often possible, effective transport is generated through precise spatiotemporal control of cargo-carrier complex assembly and disassembly. By identifying novel transient intermediates and developing kinetic models, we were able to provide direct experimental support for the presence of distinct environments at the nuclear and cytoplasmic faces of nuclear pores which are necessary for productive transport.”
Regulated nucleocytoplasmic transport via the importin superfamily of cellular transport proteins is central to cellular processes such as differentiation, proliferation and transformation, and therefore our understanding of the mechanism will increase our knowledge of these processes
This work was supported by the National Institutes of Health, the US Department of Defense, the Welch Foundation, the UK Medical Research Council and a grant from the Wellcome Trust Programme.
References
Article in PNASMurray Stewart’s group page
Siegfried Musser Laboratory, Texas A & M Health Center