 Matthew Freeman’s group in the LMB’s Cell Biology Division have discovered a novel protein, iRhom2, that plays a central role in the signalling pathways that regulate inflammation and cancer.
Matthew Freeman’s group in the LMB’s Cell Biology Division have discovered a novel protein, iRhom2, that plays a central role in the signalling pathways that regulate inflammation and cancer.
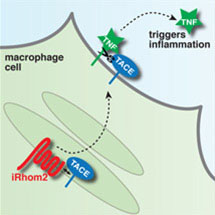
The cytokine protein, TNF, is the primary trigger of inflammation in humans. To function, active TNF must be shed from cells by the protease TACE. TACE is also responsible for the cleavage of many important growth factors, that when deregulated can contribute to cancer. Therefore, TACE acts as an important regulatory hub for signalling proteins, but up to now how TACE itself is regulated has been poorly understood.
Colin Adrain, Markus Zettl and Yonka Christova from the Freeman group, discovered that the novel protein, iRhom2, is essential for the function of TACE by showing that the loss of iRhom2 blocks inflammatory responses. They discovered that iRhom2 is needed for TACE to be trafficked from where it is made to the surface of the cell, where it is needed to activate TNF. The experiments that led to this discovery were initiated in the fruit fly Drosophila, illustrating the value of relatively simple model organisms in medical research.
Deregulated TNF activity is associated with many diseases including rheumatoid arthritis, Crohn’s disease, atherosclerosis, psoriasis, sepsis, diabetes, obesity and cancer. Drugs that block TNF function are rapidly becoming the biggest selling pharmaceuticals in the world. These drugs are primarily antibodies but they are very expensive and must be administered by injection. Attempts have been made to develop inhibitors of TACE, but because TACE controls many other signalling pathways in addition to inflammation, drug toxicity is a major problem. The work at the LMB shows that if the interaction between iRhom2 and TACE can be blocked with a drug-like molecule, this could lead to a new class of drugs that prevent TACE being transferred to the cell surface and hence reduce the production of TNF. As iRhom2 is largely restricted to inflammatory cells, this approach would cause fewer side effects than current therapies.
Further references:
Freeman Group page – now at University of Oxford
Article in Science
Perspective on Science Papers