Internal transport between different cellular compartments is a complicated process requiring formation of transport carriers, and sorting the right cargo into those carriers, for delivery to the correct part of the cell. Retromer is a protein complex that forms transport carriers departing from the cell’s central sorting station, the endosome. The architecture of the complex and how it contributes to carrier formation and cargo sorting was unknown. Researchers in John Briggs’ group in the LMB’s Structural Studies Division (and formerly at EMBL), with collaborators at the University of Queensland and the University of Cambridge have used electron cryo-tomography to reveal the structure of the retromer complex assembled on a membrane, allowing a greater understanding of how this molecular machine works.
The endosome is one of the cell’s main logistics centres. Cargoes are brought to endosomes from the cell surface or other internal compartments and sorted to other destinations to be reused or be degraded. Retromer is involved in cargo sorting at endosomes in all eukaryotes, from baker’s yeast to humans.
Retromer is composed of three subunits and can interact with proteins called sorting nexins to sort cargoes for different transport routes. Oleksiy Kovtun in John’s group, Natalya Leneva from the Universities of Queensland and Cambridge, and their collaborators at the Max Planck Institute of Biochemistry, showed for the first time the structure of the whole assembled complex interacting with a sorting nexin protein. This new model of the complex allows for a greater understanding of the ways in which the components interact with each other and also demonstrated how retromer might interact with other sorting nexin proteins to bind to different cargoes. The researchers also showed that although the function of retromer had been thought to be recognition of cargo, its primary function may actually be to remodel membranes to form tubular carriers.
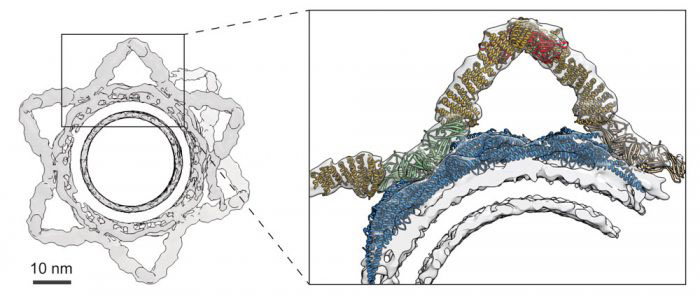
Correct functioning of the retromer complex is required for the maintenance of neurons and impaired function has been associated with neurodegenerative diseases including Parkinson’s disease and Alzheimer’s disease. This new structure and the new understanding of the role of retromer in endosomal sorting will allow scientists to better understand how mutations in the genes for retromer could lead to disease.
This work was funded by the MRC, the European Molecular Biology Laboratory, the Australian Research Council, the National Health and Medical Research Council, and the Wellcome Trust.
Further references:
Paper in Nature
John Briggs’ group page
Brett Collins’ page
David Owen’s page
The Department of Molecular Structural Biology at the Max Planck Institute of Biochemistry’s page