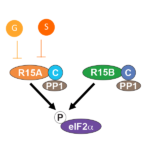
Amino acids are the building blocks of proteins. Just twenty different amino acids are strung together in different orders – like beads – to build all the proteins in living organisms. When a single type of amino acid is found consecutively within a protein, this is known as a homorepeat. Abnormal variation in the length of amino acid homorepeats has long been known to be associated with disease, such as Huntington’s.