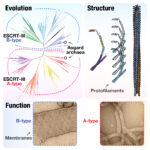
Led by postdoc Frank Bürmann, Jan Löwe’s group in the LMB’s Structural Studies Division, and Mark Dillingham’s group at the University of Bristol, have identified the mechanism the bacterial SMC complex MukBEF uses to entrap DNA ahead of loop extrusion, and found how this pathway can be inhibited by a bacteriophage protein.