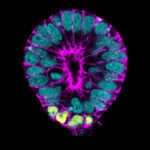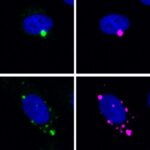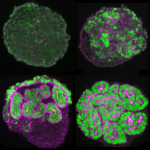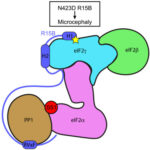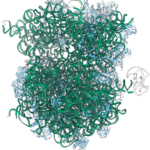
Kiarash Jamali, a Ph.D. student in Sjors Scheres’ group in the LMB’s Structural Studies Division, has designed a new machine learning software capable of fast, accurate model building at atomic levels and identification of new proteins beyond previous capabilities.