

Human brain development exhibits a number of unique characteristics, such as dramatic size expansion, unique cell types, and distinct neural stem cell behaviors. These characteristics are difficult to examine in model organisms such as mice and are often involved in neurological diseases unique to humans, like autism and schizophrenia.
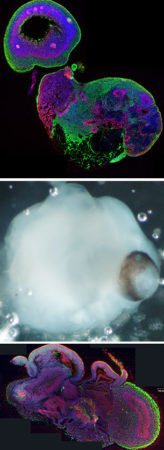
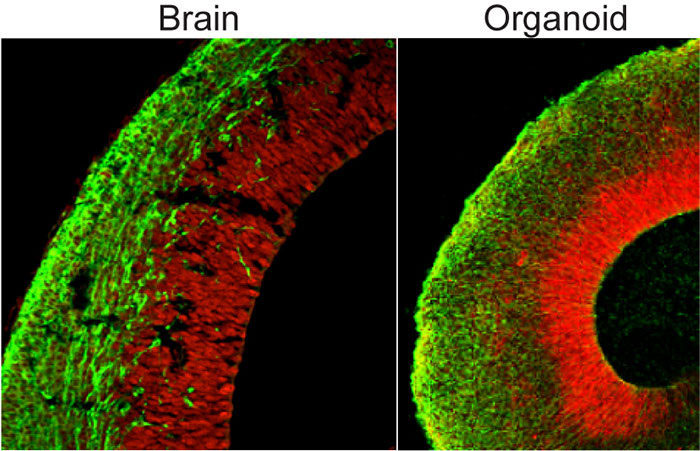
In an effort to better understand human brain development, we have developed a new model system, called cerebral organoids. Cerebral organoids, or mini-brains for short, are 3D tissues generated from human pluripotent stem cells that allow modelling of human brain development in vitro. Through a process of directed differentiation and a supportive 3D microenvironment, neural precursor tissue can spontaneously self-organize to form the stereotypic organization of the early human embryonic brain.
We have previously shown that cerebral organoids can model neurodevelopmental disorders, such as microcephaly, a disorder characterized by a significantly reduced brain size. Our current interests focus on other neurodevelopmental disorders like autism and intellectual disability, by introducing mutations seen in these disorders and examining their roles in pathogenesis in the context of organoid development. Furthermore, we are studying cellular mechanisms underlying neurodevelopmental disease progression and potential therapeutic avenues.
Finally, we are performing comparative evolutionary studies aimed at identifying conserved or unique factors in human brain size evolution, by introducing mutations to mimic the genetics of nonhuman primates and other mammals. These studies will hopefully shed light on fundamental questions about what makes us human and begin to tackle difficult questions pertaining to a number of debilitating neurological diseases.
Selected Papers
- Chiaradia I, Imaz-Rosshandler I, Nilges BS, Boulanger J, Pellegrini L, Das R, Kashikar ND, Lancaster MA. (2023)
Tissue morphology influences the temporal program of human brain organoid development
Cell Stem Cell 30(10): 1351-1367 - Kelava I., Chiaradia I., Pellegrini L., Kalinka A. T., Lancaster M. A. (2022)
Androgens increase excitatory neurogenic potential in human brain organoids.
Nature 602(7895): 112-116 - Benito-Kwiecinski S, Giandomenico SL, Sutcliffe M, Riis ES, Freire-Pritchett P, Kelava I, Wunderlich S, Martin U, Wray GA, McDole K, Lancaster MA. (2021)
An early cell shape transition drives evolutionary expansion of the human forebrain.
Cell S0092-8674(21): 00239-7. doi: 10.1016/j.cell.2021.02.050. Online ahead of print. - Pellegrini L, Albecka A, Mallery DL, Kellner MJ, Paul D, Carter AP, James LC, Lancaster MA. (2020)
SARS-CoV-2 Infects the Brain Choroid Plexus and Disrupts the Blood-CSF Barrier in Human Brain Organoids.
Cell Stem Cell 27(6): 951-961.e5. doi: 10.1016/j.stem.2020.10.001. Epub 2020 Oct 13. - Pellegrini L, Bonfio C, Chadwick J, Begum F, Skehel M, Lancaster MA. (2020)
Human CNS barrier-forming organoids with cerebrospinal fluid production.
Science 369(6500): eaaz5626 - Giandomenico SL, Mierau SB, Gibbons GM, Wenger LMD, Masullo L, Sit T, Sutcliffe M, Boulanger J, Tripodi M, Derivery E, Paulsen O, Lakatos A, Lancaster MA. (2019)
Cerebral organoids at the air-liquid interface generate diverse nerve tracts with functional output.
Nat Neurosci. 22(4): 669-679. - Lancaster MA, Corsini NS, Wolfinger S, Gustafson EH, Phillips AW, Burkard TR, Otani T, Livesey FJ, Knoblich JA. (2017)
Guided self-organization and cortical plate formation in human brain organoids.
Nat Biotechnol. 35(7): 659-666. - Lancaster, M.A., Renner, M., Martin, C.A., Wenzel, D., Bicknell, L.S., Hurles, M.E., Homfray, T., Penninger, J.M., Jackson, A.P. and Knoblich, J.A. (2013)
Cerebral organoids model human brain development and microcephaly.
Nature 501: (7467):373-379.
Group Members
- Alexander Anderson
- José González Martínez
- Luca Guglielmi
- Alexander Justin
- Nuzli Karam
- Wanda Kukulski
- Feline Lindhout
- Daniel Lloyd-Davies-Sánchez
- Aly Makhlouf
- Miguel Mestre
- Charlie Morris
- Magdalena Sutcliffe (Barczyk)
- Hanna Szafranska
- Steven Wingett